临床上,IUA的治疗包括以下几种方式:①药物治疗,通常采用雌、孕激素序贯治疗促进子宫内膜的生长和修复,但治疗周期长且不适用于所有类型的IUA患者;②宫腔镜下宫腔粘连分离术(transcervical resection of adhesion,TCRA),通过手术方式分离粘连的宫腔组织,恢复子宫内膜的正常结构和功能,以改善月经周期和生育能力,然而,手术分离存在再次粘连的风险;③放置球状宫腔支架分隔宫腔壁防止粘连,放置1个月后取出仍存在复发风险,会给患者带来额外负担。针对宫腔支架需二次手术取出的问题,水凝胶支架因其良好的生物相容性和可降解性备受关注。使用水凝胶支架植入粘连部位可为子宫前后壁提供机械支撑,显著降低了TCRA术后IUA的发生率[4]。然而,传统水凝胶支架仅依赖其负载的药物达到短暂的抗炎效果,对妊娠改善作用有限。子宫自身的挤压可能会导致植入的水凝胶从宫腔流出,无法持续发挥作用。此外,水凝胶支架缺乏个性化定制,难以满足不同患者的需求。
近年来,3D生物打印技术在促进组织修复与再生中取得重要进展。针对目前IUA治疗存在的问题,利用3D生物打印技术将水凝胶制备成与损伤部位互补的形态结构,使其能在子宫损伤部位长期停留,促进生理功能的恢复,这项技术有望为IUA的治疗带来新的突破及可能性。
1 IUA的病理生理机制
IUA主要是由子宫内膜损伤后过度纤维化导致。当子宫内膜的基底层受损时,其自我修复机制失效,内膜无法再生。过度的炎症反应及成纤维细胞的异常增生导致子宫内膜瘢痕形成。在这些瘢痕中,大量的胶原纤维替代了正常的内膜结构,并在宫腔前后壁形成纤维结缔组织连接,导致粘连。
对IUA的发生机制,目前有两种观点:一种观点认为,子宫内膜基底层含有少量内膜上皮干细胞和间充质干细胞(mesenchymal stem cells,MSCs),它们对子宫内膜腺体和基质的周期性再生具有一定促进作用[5]。然而,宫腔手术会导致这些干细胞数量减少,妨碍了子宫内膜腺体和基质的有效再生。而且宫腔手术会使成纤维细胞激活且大量增殖,产生的胶原蛋白引发内膜表面过度纤维化,最终导致IUA的形成。另外一种观点认为子宫内膜纤维化是细胞因子导致的,如转化生长因子-β1(transforming growth factor-β1,TGF-β1)会刺激胶原蛋白的合成,通过TGF-β1/Smad信号传导途径促进纤维化进程。TGF-β1激活其受体后,进而激活下游信号分子Smad2和Smad3,磷酸化后的Smad3能促进纤维细胞内Ⅰ型胶原蛋白等基质蛋白的合成,从而加速胶原的沉积和纤维化进程[6]。这些研究揭示了IUA发生的细胞和分子机制,为未来IUA治疗提供了重要线索。
2 3D打印水凝胶生物支架
2.1 3D打印生物墨水的选择
3D打印生物墨水是指用于3D打印的可构建组织或器官结构的材料。这些生物墨水通常包含特定的细胞及支持细胞生长的基质材料,这些材料需具备以可控的方式进行3D打印的特性并且体内移植后不会引起免疫排斥反应。目前用于3D打印的生物墨水通常为水凝胶材料,包括明胶(gelatin,Gel)、透明质酸(hyaluronic acid,HA)和海藻酸钠(sodium alginate,SA)等。
Gel是一种来自动物胶原蛋白的水溶性蛋白质,利于细胞黏附和生长,具备优异的生物相容性。Gel可通过调整温度来实现水凝胶状态和溶液状态的转变,适用于多种细胞的生物打印[7]。甲基丙烯酰化明胶(gelatin methacrylate,GelMA)通过在Gel中引入丙烯酰基,使得GelMA具备光交联特性。GelMA的凝胶化速度和强度可通过光交联进行调控。这意味着可以使用紫外光或蓝光照射来控制GelMA凝胶的形成,从而在3D打印过程中实现精确的控制。
HA是一种天然存在于人体的多糖,是细胞外基质重要组成成分,在3D生物打印中,HA可以作为生物墨水的一部分,提供细胞所需的支持结构[8]。在HA分子的羟基上引入甲基丙烯酰基团(hyaluronic acid methacrylate,HAMA),从而赋予HA光交联性和稳定性。通过化学改性的方法,使其更适用于3D生物打印。子宫内膜中存在HA受体,选用HA水凝胶是治疗IUA理想的生物墨水。
目前多种水凝胶材料已被广泛应用于IUA的治疗研究,常见的水凝胶材料使用参数及其各自的优缺点见表1。
表1 水凝胶材料参数及其优缺点
| 序号 | 水凝胶材料 | 参数 | 优点 | 缺点 |
|---|---|---|---|---|
| 1 | GelMA、SA[9] | 10%GelMA,2%SA,1%光引发剂 | 多孔性、可压缩性高、生物相容性高 | GelMA体内降解较慢、单独支架促修复效果有限 |
| 2 | Gel、SA[10] | 20%Gel和4%SA,2∶1体积比,4度打印,10%氯化钙交联 | 可在体外体内长期支持负载细胞的活性,体内可自然降解,生物相容性高 | 人的诱导性多能干细胞移植到大鼠存在免疫反应,子宫内膜修复疗效有限 |
| 3 | 自交联HA[11] | 5 mg/mL | 为UC-MSCs移植提供载体,防止细胞流出,提供物理支持 | 单独支架移植未改善恒河猴的宫腔形态及月经周期 |
| 5 | Ⅰ型胶原蛋白[12] | 0.5%胶原冻干,105 ℃热脱水15 h | 具有细胞外基质同样的纤维结构、具有多孔结构,适合MSCs的负载黏附和营养支持 | 需要和干细胞联合使用才能达到促修复和改善妊娠效果 |
| 6 | 壳聚糖[13] | 2%壳聚糖/乙酸溶液3 mL加入1.5 mL交联液,搅拌15 min,37 ℃交联形成水凝胶 | 生物相容性高,具备抗菌性能,稳定性高 | 制备过程较为复杂,氨基成分易被化学修饰,机械强度弱 |
注:UC-MSCs脐带间充质干细胞(umbilical cord mesenchymal stem cells)。
2.2 3D生物打印机
与传统的3D打印机有所不同,3D生物打印机需要在打印过程中保持生物相容性、细胞存活性和可控性。按照原理不同,现阶段常用的3D生物打印机分为喷墨式、挤出式和立体光刻式。
喷墨式3D打印机通过电压或热力作用使打印材料以液滴的形式从喷嘴中喷出,在收集板上交联堆积从而获得3D模型。Xu等[14]使用热喷墨式3D打印机制备负载人羊水干细胞的海藻酸盐/胶原蛋白支架,负载细胞活性超过90%,证明喷墨式打印过程对细胞活性影响较小。但由于驱动压力较小,要求打印材料需具有较小的黏度,因而该打印机不适用于高黏性生物墨水的使用。
挤出式生物打印机源自喷墨式打印,由于其多功能性和经济性,是使用最广泛的3D生物打印机。通过对打印材料施加持续的作用力,在尖嘴处产生连续的细丝。这种方法可在不同部位打印不同的生物材料及细胞,可在体外构建多种细胞空间排列模型[15]。使用挤出式打印机可打印负载不同细胞的双层生物支架,符合子宫内膜的正常生理结构。但需要注意的是,挤压时产生的剪切力过大会导致细胞破碎,应严格控制材料的黏度及挤出压力从而提高负载细胞的存活率。同时该方法由于挤出的细丝依赖于针头的粗细,适用于毫米级支架的制备。打印较小尺寸的模型时容易失真,不适用于微米级别精密支架的制备。
立体光刻式3D打印运用光诱导聚合原理,当打印材料暴露在紫外线或可见光下时,掺在打印材料中的光引发剂会被诱导发生聚合反应,从而使特定位置材料按照程序光交联固化成型[16]。该打印方式具有精度高、速度快的优势,但需要生物墨水具有光交联特性例如常用的GelMA及HAMA等。同时,生物墨水中,掺入的某些光引发剂会产生细胞毒性反应及紫外线照射易致细胞突变,影响细胞的正常发育。目前使用蓝光405 nm交联及LAP光引发剂改进后可显著降低细胞毒性以降低上述不利影响。
不同打印方式在打印精度、细胞或药物的活性等方面各有优势,故应根据子宫内膜损伤程度及打印材料等综合考虑来选择合适的打印方式。如考虑负载多种细胞进行IUA支架打印,选择挤出式打印方式;若需打印精度高的支架材料,则选择立体光刻式3D打印。
3 3D打印水凝胶支架在IUA防治中的应用
3.1 3D打印水凝胶支架优势
理想的3D打印防粘连支架包括以下特征:①支架形状与子宫腔匹配良好;②有足够的有效面积隔离子宫前后壁,甚至可延伸至宫颈管内,以预防子宫下段及宫颈管粘连;③支撑力度适宜,防止子宫内膜受压缺血坏死;④为满足各种尺寸宫腔的需要而设计的各种尺寸。3D打印水凝胶支架可根据宫腔尺寸进行定制设计,打印合适尺寸的支架以分隔子宫前后壁,防止子宫损伤后粘连带的形成。
3.2 原位药物递送
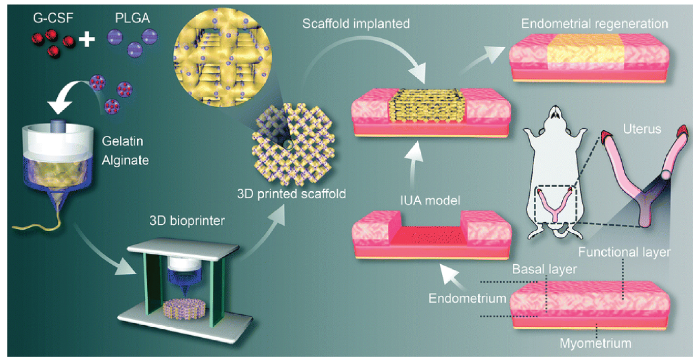
3D打印水凝胶支架可负载促进子宫内膜修复的药物及生长因子促进IUA修复,例如在支架植入体内后,负载药物随着水凝胶的自降解后局部释放,实现TCRA术后在损伤局部持续精准释放。Wen等[17]使用聚乳酸-羟基乙酸共聚物(polylactic-co-glycolic acid,PLGA)微球负载粒细胞集落刺激因子(granulate colony-stimulating factor,G-CSF),建立缓释微球(sustained release microsphere,SRM)系统(G-CSF-SRM),实现G-CSF的持续释放。将G-CSF-SRM、20%的Gel溶液及4%的SA溶液以体积比2∶1∶1混合均匀,经挤出式3D打印机打印支架后,植入大鼠IUA模型缺损处,在损伤局部实现G-CSF缓释(见图1)。结果显示,移植5周后,3D打印支架+微球组的子宫内膜厚度[(589±70)μm vs. (242±73)μm,P<0.01]和腺体数量[(7.5±1.1) vs. (2.5±0.9),P<0.01]均显著高于仅用磷酸盐缓冲液(phosphate buffered saline,PBS)治疗的IUA组,胶原蛋白含量高于仅用PBS治疗的IUA组[(85.1±3.1)% vs. (51.6±6.1)%,P<0.05],表明3D打印支架+微球组的子宫内膜结构再生程度较优。该研究还通过测定大鼠的妊娠率以及大鼠怀孕18 d后手术部位的胚胎数量来评估修复后子宫内膜的妊娠功能恢复情况,结果显示,3D打印支架+微球组妊娠率(75% vs. 17%,P<0.05)和胚胎数量(5.4个 vs. 1.5个,P<0.05)显著高于仅用PBS治疗的IUA组。由此推测,3D打印的生物活性支架可促进子宫内膜结构和功能重建,从而恢复受损子宫内膜的妊娠功能。负载药物的3D打印支架主要利用3D打印支架的优势(如3.1章节所述),同时在损伤局部释放负载的药物,从而起到更好的疗效。
图1
图1
3D打印水凝胶支架缓释G-CSF体内促大鼠IUA模型修复的示意图[17]
注:G-CSF粒细胞集落刺激因子;PLGA聚乳酸-羟基乙酸共聚物;Gelatin Alginate明胶海藻酸钠;3D Bioprinter 3D生物打印机;3D printed scaffold 3D打印支架;Scaffold implanted支架移植;IUA model宫腔粘连模型;Endometrial regeneration子宫内膜再生;Uterus子宫;Basal layer 子宫基底层;Functional layer子宫功能层;Endometrium子宫内膜层;Myometrium子宫肌层。
3.3 原位干细胞递送
干细胞向病变部位的归巢和迁移在组织再生中发挥重要作用[18]。研究表明,与健康女性相比,IUA患者干细胞的迁移和侵袭能力明显降低,这可能间接影响受损子宫内膜的自我修复能力。此外,当子宫内膜严重受损时,子宫内膜干细胞的减少可能影响子宫内膜的再生[19]。因此,自体和异体干细胞移植可能对IUA的治疗有效[20]。Zheng等[21]将荧光标记的人脐带间充质干细胞(umbilical cord mesenchymal stem cells,UC-MSCs)注射到SD大鼠体内,结果显示其具有分化为上皮细胞、血管内皮细胞和雌激素受体细胞的能力,但移植的UC-MSCs易流失。Xu等[22]构建了UC-MSCs的胶原支架并将其移植到大鼠子宫瘢痕模型中,移植后第60天,支架/UC-MSCs组每个子宫角的胚胎总数[5.50(0~8)个]显著高于PBS组[1.50(0~6)个,P<0.01]、支架组[2.50(0~6)个,P<0.05]和UC-MSCs组[1.50(0~7)个,P<0.05]。结果表明,UC-MSCs通过上调基质金属蛋白酶-9(matrix metalloproteinase-9,MMP-9)的分泌促进子宫瘢痕中胶原蛋白的降解,并促进子宫内膜、子宫肌层和血管再生,从而改善生育能力。2020年Wang等[11]的研究表明,UC-MSCs/HA-Gel支架移植恒河猴IUA模型,发现负载UC-MSCs的支架比仅放置支架效果更好。UC-MSCs/HA-Gel复合物可部分修复机械损伤引起的IUA,其具有可靠的抗粘连性能,并能促进子宫内膜变厚、纤维化形成量减少、子宫内膜腺体数量增加等。目前负载人UC-MSCs的胶原支架治疗复发性IUA已进入Ⅰ期临床试验,结果提示,与治疗前相比,术后3个月的子宫内膜平均最大厚度增加,IUA评分降低;在30个月的随访结束时,26例患者中有10例妊娠[23]。
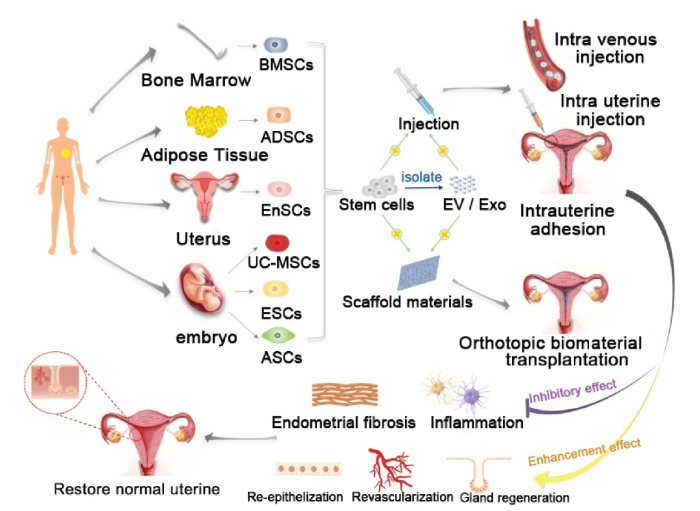
目前治疗IUA的研究主要涉及胚胎干细胞、成体干细胞及诱导性多能干细胞等。胚胎干细胞增殖能力强,但有成瘤风险,应用受限。成体干细胞恶性潜能较低,常被应用于干细胞治疗。MSCs是中胚层早期形成的多能成体干细胞,具备多能分化、高度增殖潜能和免疫调节功能,且来源广泛,可从脐带、子宫内膜息肉、月经血、骨髓和脂肪组织等多种组织中分离获得。目前,骨髓间充质干细胞(bone marrow mesenchymal stem cells,BMSCs)、UC-MSCs、脂肪间充质干细胞(adipose derived mesenchymal stem cells,ADSCs)等均在IUA的治疗研究中起到促修复作用(见图2)。这些干细胞通过旁分泌作用,上调或下调各种细胞信号通路的表达,对子宫内膜的厚度和腺体数量、血管重塑、抑制过度纤维化和炎症反应发生方面起到一定作用[21⇓⇓-24],从而达到恢复子宫内膜的正常组织结构,改善生育功能的目的[25]。
图2
图2
应用MSCs修复受损子宫内膜的方法及机制[5]
注:Bone Marrow骨髓;BMSCs骨髓间充质干细胞;Adipose Tissue脂肪组织;ADSCs脂肪间充质干细胞;Uterus子宫;EnSCs(Endometrial stem/progenitor cells)子宫内膜干细胞/祖细胞;embryo胚胎;UC-MSCs脐带间充质干细胞;ESCs (Embryonic stem cells)胚胎干细胞;ASCs(Amniotic membrane stem cells)羊膜干细胞;Stem cells干细胞;isolate分离;Injection注射;EV/Exo囊泡/外泌体;Scaffold materials支架材料;Intravenous injection静脉注射;Intrauterine injection子宫内注射;Intrauterine adhesion宫腔粘连;Orthotopic biomaterial transplantation原位生物材料移植;Inhibitory effect抑制作用;Enhancement effect促进作用;Inflammation炎症;Endometrial fibrosis子宫内膜纤维化;Gland regeneration腺体再生;Revascularization血管再生;Re-epithelization再上皮化;Restore normal uterine 恢复正常子宫。
将水凝胶负载干细胞移植至子宫内膜粘连处,干细胞宫腔内留存率低。通过3D打印出与宫腔互补的支架负载干细胞原位移植后不易流失,从而在损伤局部发挥干细胞的功能达到改善妊娠的目的[26]。例如Ji等[10]利用挤出式生物打印机制备负载人诱导性多能干细胞来源的间充质干细胞(human induced pluripotent stem cell derived mesenchymal stem cells,hiMSCs)的Gel支架。与单独注射hiMSCs组相比,3D打印的hiMSCs支架移植到大鼠IUA损伤处治疗后,大鼠妊娠率明显提高(60% vs. 33%,P<0.05)。Feng等[27]利用3D打印技术构建GelMA和甲基丙烯酸胶原(collagen methacrylate,ColMA)支架负载人羊膜间充质干细胞(human amniotic mesenchymal stem cells,hAMSCs)用于IUA治疗。结果表明,与单独的GelMA水凝胶相比,1%的ColMA具备最适机械性能,即增加了压缩模量(43.63±2.9 vs. 24.7±7.7,P<0.05),降低了溶胀比率(20.9% vs.25.2%,P<0.05),这有利于防止过度溶胀导致的宫腔损伤。3D打印的GelMA/ColMA支架中hAMSCs形态上表现出伸长和增粗的肌动蛋白丝,增殖速率快,表明GelMA/ColMA支架有利于hAMSCs移植。将3D打印的GelMA/ColMA/hAMSC支架移植大鼠IUA模型可防止粘连发生,通过改善胶原沉积、减少纤维化面积,促进大鼠子宫内膜修复。
然而,将干细胞简单地与水凝胶混合的方式进行打印,无法为负载细胞提供可控的孔隙形状,这影响了细胞的存活、增殖、迁移和分化以及临床应用中的可重复性。为解决这一问题,Lu等[28]使用聚环氧乙烷(Polyethylene oxide,PEO)作为制孔剂,制备具有可控微米级孔的3D打印GelMA支架(PH组)。该支架具有良好的可注射性,在压缩和注射后可恢复至原始形状。这些功能不仅有利于ADSCs递送,还能以微创的方式将支架注射到子宫内膜缺陷处,提升了临床应用性。将ADSCs接种到具有微孔结构的3D打印支架(ADSC@PH)中,其对人血管内皮细胞(human vascular endothelial cells,HUVECs)的迁移、增殖和血管形成有促进作用。将ADSC@PH植入大鼠IUA模型后,其可通过抑制炎性因子的分泌及提高抗炎巨噬细胞比例达到抑制炎症的效果。移植14 d后ADSC@PH组的微血管密度(23.8±1.3)高于其他组(IUA模型组:4.8±0.4,PH组:4.4±0.5,ADSC组:14.0±3.5,EXO组:8.8±0.8,EXO@PH组:16.6±2.5,P<0.001),表明ADSC@PH可以稳定、长期地增强新生血管形成。ADSC@PH组的子宫内膜厚度[(792±36.5)μm vs.(217±31.1)μm,P<0.001]和腺体数量[(25±1) vs. (7±2),P<0.001]显著高于IUA模型组,表明这种3D打印的多孔支架适用于干细胞递送,对子宫内膜的修复及IUA治疗具有一定疗效。
综上,利用3D打印支架负载和移植干细胞后可以防止粘连形成,改善子宫内膜形态,促进子宫内膜血管再生,增加子宫内膜厚度,提高妊娠率。通过对打印生物墨水的组成成分和支架内部结构的优化可进一步提升干细胞的治疗效果,从而达到无创植入、改善妊娠结局的目的。目前应用水凝胶材料负载MSCs移植的研究已广泛开展,但使用3D打印水凝胶生物支架移植干细胞的研究仍处于起步阶段,随着生物材料的开发及3D生物打印技术的日益成熟,将会成为下一阶段IUA治疗的研究重点。
3.4 原位外泌体递送
近年来,MSCs来源的外泌体作为一种含有生物活性分子的小囊泡,在组织修复和再生中显示出潜在的重要作用。其包含的多种生物小分子,如mRNA、微小RNA(microRNA,miRNA)、蛋白质和脂质等对于细胞增殖、血管生成、炎症调节等过程至关重要。通过外泌体释放的生物活性物质尤其是miRNA,有望减少IUA瘢痕组织的形成,改善内膜再生,并促进正常组织的恢复。大量临床前研究表明,外泌体在治疗IUA中的作用机制涉及3个方面:①MSCs来源的外泌体可通过激活Wnt/β-连环蛋白(β-catenin)信号通路调节Bcl-2和Bax的表达,促进细胞增殖、抑制细胞凋亡[29];②外泌体内含抗炎成分具有炎症调节作用,作用到靶细胞以抑制炎症反应[30];③外泌体能促进受损内膜基底层新生血管的形成,特别是通过miRNA(如miR-29b)促进血管生成相关基因的表达,例如血管内皮生长因子(vascular endothelial growth factor,VEGF)和成纤维细胞生长因子(fibroblast growth factor,FGF)基因[31]。
MSCs及MSCs来源的外泌体治疗IUA具有相似的生物功能,例如BMSCs来源的外泌体可通过miR-223-3p抑制白细胞介素-18(interleukin-18,IL-18)和IL-1β炎症因子释放,能缓解IUA的症状[32];ADSCs来源的外泌体则通过促进整合素β3、IL及VEGF等物质的生成,促进胶原的重塑助于恢复子宫结构[33];UC-MSCs来源的外泌体则展现出抗炎和抗纤维化的效果,与雌激素联合应用时增强子宫内膜再生作用[34]。这些研究表明,作为一种新兴的、基于干细胞的外泌体疗法能有效规避移植MSCs可能带来的免疫排斥、肿瘤形成和血栓等风险。因此,未来通过3D打印技术原位递送干细胞外泌体的非细胞治疗方法是一种有前景的修复手段。
4 结语与展望
3D生物打印作为一种创新的生物医学技术,尽管目前在这一领域的研究尚处于早期阶段,但其在未来对IUA治疗具有重要潜力,包括①定制个性化治疗方案:通过3D生物打印技术,根据患者的具体情况和需求,定制个体化生物材料和支架,用于修复和分离受损的子宫内膜,为患者提供个性化治疗方案;②精确重建子宫内膜:3D生物打印可根据患者受损的子宫内膜结构定制互补的类内膜结构,通过打印生物墨水和细胞,支持内膜组织的再生和生长,防止宫腔再次粘连的发生;③定向药物递送系统:利用生物打印技术制造的微型支架可作为药物递送系统,可将药物或生长因子定向释放到特定的治疗区域,促进组织修复和再生;④干细胞及干细胞外泌体治疗:通过将患者自身的干细胞体外提取和扩增获取外泌体,将外泌体负载到3D打印的支架中原位移植到损伤区域,可促进组织生长和修复,并提高治疗效果。
随着高分子材料的发展,合成新型适宜IUA治疗的3D打印生物墨水仍然是研究重点。需寻找生物相容性良好、能够支持细胞生长的材料,同时具有适当的力学特性与适宜的降解特性,能够在植入后长期支撑和维持修复的效果;与此同时,需开发高精度的3D打印机实现高精度快速打印,在打印过程中确保细胞的活性及功能,保证打印出的组织结构能够在体内发挥有效作用;进行严格的临床试验和验证,确保3D生物打印在IUA治疗中的安全性和有效性。尽管目前面临一些挑战,但随着生物打印技术的不断发展和完善,以及科学研究的不断深入,3D生物打印有望成为治疗IUA的有力工具,为患者提供更加个性化和有效的治疗方案。
参考文献
Reproductive performance of women with and without intrauterine adhesions following recurrent dilatation and curettage for miscarriage: long-term follow-up of a randomized controlled trial
[J].
Focus on the Primary Prevention of Intrauterine Adhesions: Current Concept and Vision
[J].
A Lancet Commission on 70 years of women′s reproductive, maternal, newborn, child, and adolescent health in China
[J].
Effectiveness of Hyaluronic Acid Gel and Intrauterine Devices in Prevention of Intrauterine Adhesions after Hysteroscopic Adhesiolysis in Infertile Women
[J].
Stem cell-based therapy for ameliorating intrauterine adhesion and endometrium injury
[J].
Transforming growth factor-β1 in intrauterine adhesion
[J].
Recent Advances on Bioprinted Gelatin Methacrylate-Based Hydrogels for Tissue Repair
[J].
Manufacturing of self-standing multi-layered 3D-bioprinted alginate-hyaluronate constructs by controlling the cross-linking mechanisms for tissue engineering applications
[J].
Porous scaffolds from droplet microfluidics for prevention of intrauterine adhesion
[J].Severe intrauterine adhesions (IUAs) have a great negative impact on women's psychological and reproductive health. It remains a significant challenge to prevent postoperative IUAs because of the complications of various clinical preventive measures and incompatibility of uterine cavity morphology. Herein, we present a new drug-loadedporous scaffold based on a microfluidic droplet template, which combines the characteristics of the artificial biocompatible material GelMA and the natural polysaccharide material Na-alginate. By changing the containers that collect the microfluidic droplets, the porous scaffold conforming to the shape of the uterine cavity could be obtained. The porous structure, mechanical property, and flexibility impart the scaffold with compressibility and send it to the uterus through the vagina. In addition, the external-internal connected open structures could load and control the release of drugs to repair the damaged region continuously in vivo. To verify the antiadhesion and repair of drug-loaded porous scaffolds, we tested the system in the rat model of IUAs, and it was demonstrated that the system had the ability to improve neovascularization, cellularize the damaged tissue, and repair the endometrium. These features provide the drug-loaded porous scaffolds with new options for the improvement of postoperative IUAs. STATEMENT OF SIGNIFICANCE: Intrauterine adhesions are caused by various causes of damage to the endometrial basal layer, thus leading to part or entire adhesions in the cervical or uterine cavity. Clinically, various preventive measures reach the barrier effect through the physical barrier, which are difficult to further promote the repair of the damaged endometrium, and most of them have apparent side effects. This study aims to prepare compressible and biodegradable three-dimensional porous drug-loading biological scaffolds. GelMA and Na-alginate have desirable biocompatibility. The interconnect porous scaffolds, which were prepared through the combination of biomaterials and single emulsion microfluidics, not only have compressibility but also provide space for drug delivery and release. This system can further promote the repair of the endometrium while preventing adhesion.Copyright © 2018 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
3D Bioprinting a human iPSC-derived MSC-loaded scaffold for repair of the uterine endometrium
[J].Common events in the clinic, such as uterine curettage or inflammation, may lead to irreversible endometrial damage, often resulting in infertility in women of childbearing age. Currently, tissue engineering has the potential to achieve tissue manipulation, regeneration, and growth, but personalization and precision remain challenges. The application of "3D cell printing" is more in line with the clinical requirements of tissue repair. In this study, a porous grid-type human induced pluripotent stem cell-derived mesenchymal stem cell (hiMSC)-loaded hydrogel scaffold was constructed using a 3D bioprinting device. The 3D-printed hydrogel scaffold provided a permissive in vitro living environment for hiMSCs and significantly increased the survival duration of transplanted hiMSCs when compared with hiMSCs administered locally in vivo. Using an endometrial injury model, we found that hiMSC transplantation can cause early host immune responses (the serological immune response continued for more than 1 month, and the local immune response continued for approximately 1 week). Compared with the sham group, although the regenerative endometrium failed to show full restoration of the normal structure and function of the lining, implantation of the 3D-printed hiMSC-loaded scaffold not only promoted the recovery of the endometrial histomorphology (endometrial tissue and gland regeneration) and the regeneration of endometrial cells (stromal cells and epithelial cells) and endothelial cells but also improved endometrial receptivity functional indicators, namely, pinopode formation and leukemia inhibitory factor and αvβ3 expression, which partly restored the embryo implantation and pregnancy maintenance functions of the injured endometrium. These indicators were significantly better in the 3D-printed hiMSC-loaded scaffold group than in the unrepaired (empty) group, the hiMSCs alone group and the 3D scaffold group, and the empty group showed the worst repair results. Our study confirm that the 3D-printed hiMSC-loaded hydrogel scaffold may be a promising material for endometrial repair.Copyright © 2020. Published by Elsevier Ltd.
In situ repair abilities of human umbilical cord-derived mesenchymal stem cells and autocrosslinked hyaluronic acid gel complex in rhesus monkeys with intrauterine adhesion
[J].
A collagen scaffold loaded with human umbilical cord-derived mesenchymal stem cells facilitates endometrial regeneration and restores fertility
[J].In women of reproductive age, severe injuries to the endometrium are often accompanied by endometrial scar formation or intrauterine adhesions (IUAs), which can result in infertility or miscarriage. Although many approaches have been used to treat severe IUAs, high recurrence rates and endometrial thinning have limited therapeutic efficiency. In this study, a collagen scaffold (CS) loaded with human umbilical cord-derived mesenchymal stem cells (UC-MSCs) was fabricated and applied for endometrial regeneration. The CS/UC-MSCs promoted human endometrial stromal cell proliferation and inhibited apoptosis in vitro through paracrine effects. In a model of endometrial damage, transplantation with the CS/UC-MSCs maintained normal luminal structure, promoted endometrial regeneration and collagen remodeling, induced intrinsic endometrial cell proliferation and epithelium recovery, and enhanced the expression of estrogen receptor α and progesterone receptor. An improved ability of the regenerated endometrium to receive embryos was confirmed. Together, our results indicate that the CS/UC-MSCs promoted endometrial structural reconstruction and functional recovery. Topical administration of the CS/UC-MSCs after trans-cervical resection of adhesions might prevent re-adhesion, promote endometrium regeneration and improve pregnancy outcomes for patients with severe IUAs. STATEMENT OF SIGNIFICANCE: Intrauterine adhesions due to severe endometrium injuries happen frequently in clinic and become one of the crucial reasons for women's infertility or miscarriage. Therefore, how to regenerate the damaged endometrium is a big challenge. In this study, a collagen scaffold (CS) loaded with human umbilical cord-derived mesenchymal stem cells (UC-MSCs) was fabricated and applied for endometrium regeneration. Herein, UC-MSCs, known for low immunogenicity and high proliferative potential, exhibit promising potential for endometrium regeneration; and collagen scaffolds provide suitable physical support. It was proved that transplantation with CS/UC-MSCs promoted endometrial regeneration and fertility restoration. It suggested that topical administration of CS/UC-MSCs in uterus could be a promising strategy for patients suffering severe intrauterine adhesion and infertility.Copyright © 2019 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
Controlled releasing of SDF-1α in chitosan-heparin hydrogel for endometrium injury healing in rat model
[J].Herein we report a facile approach for chitosan-heparin hydrogels with controlled release manner and their applications for intrauterine adhesion. The sol precursor was converted to gel at physiological temperature in 15 min. FTIR, SEM and swelling test were performed to characterize their compositions, morphologies and stability. In vitro releasing profiles was investigated in PBS solutions. Intrauterine injured rat model was established and treated with different methods. The results of H&E staining, Masson trichrome staining, western blots assay, immunohistochemical staining and immunofluorescence staining revealed that endogenous c-kit positive stem cells (HSCs) were recruited to the injury site and promoted the wound recovery. After 7 days' treatment, uterus treated with SDF-1α releasing hydrogel showed no difference with control group on endometrial thickness, glands number and fibrosis level. This work provides a possible method for intrauterine adhesion healing.Copyright © 2019 Elsevier B.V. All rights reserved.
Inkjet printing of viable mammalian cells
[J].The purpose of this study was to explore the use of a commercial thermal printer to deposit Chinese Hamster Ovary (CHO) and embryonic motoneuron cells into pre-defined patterns. These experiments were undertaken to verify the biocompatibility of thermal inkjet printing of mammalian cells and the ability to assemble them into viable constructs. Using a modified Hewlett Packard (HP) 550C computer printer and an HP 51626a ink cartridge, CHO cells and rat embryonic motoneurons were suspended separately in a concentrated phosphate buffered saline solution (3 x). The cells were subsequently printed as a kind of "ink" onto several "bio-papers" made from soy agar and collagen gel. The appearance of the CHO cells and motoneurons on the bio-papers indicated an healthy cell morphology. Furthermore, the analyses of the CHO cell viability showed that less than 8% of the cells were lysed during printing. These data indicate that mammalian cells can be effectively delivered by a modified thermal inkjet printer onto biological substrates and that they retain their ability to function. The computer-aided inkjet printing of viable mammalian cells holds potential for creating living tissue analogs, and may eventually lead to the construction of engineered human organs.Copyright 2004 Elsevier Ltd.
Progress in 3D bioprinting technology for tissue/organ regenerative engineering
[J].
A review on stereolithography and its applications in biomedical engineering
[J].Stereolithography is a solid freeform technique (SFF) that was introduced in the late 1980s. Although many other techniques have been developed since then, stereolithography remains one of the most powerful and versatile of all SFF techniques. It has the highest fabrication accuracy and an increasing number of materials that can be processed is becoming available. In this paper we discuss the characteristic features of the stereolithography technique and compare it to other SFF techniques. The biomedical applications of stereolithography are reviewed, as well as the biodegradable resin materials that have been developed for use with stereolithography. Finally, an overview of the application of stereolithography in preparing porous structures for tissue engineering is given.2010 Elsevier Ltd. All rights reserved.
3D-printed hydrogel scaffold-loaded granulocyte colony-stimulating factor sustained-release microspheres and their effect on endometrial regeneration
[J].
Phenotype and biological characteristics of endometrial mesenchymal stem/stromal cells: A comparison between intrauterine adhesion patients and healthy women
[J].
Human umbilical cord-derived mesenchymal stem cells prevent the progression of early diabetic nephropathy through inhibiting inflammation and fibrosis
[J].Diabetic nephropathy (DN) is one of the most serious complications of diabetes and the leading cause of end-stage chronic kidney disease. Currently, there are no effective drugs for treating DN. Therefore, novel and effective strategies to ameliorate DN at the early stage should be identified. This study aimed to explore the effectiveness and underlying mechanisms of human umbilical cord mesenchymal stem cells (UC-MSCs) in DN.We identified the basic biological properties and examined the multilineage differentiation potential of UC-MSCs. Streptozotocin (STZ)-induced DN rats were infused with 2 × 10 UC-MSCs via the tail vein at week 6. After 2 weeks, we measured blood glucose level, levels of renal function parameters in the blood and urine, and cytokine levels in the kidney and blood, and analyzed renal pathological changes after UC-MSC treatment. We also determined the colonization of UC-MSCs in the kidney with or without STZ injection. Moreover, in vitro experiments were performed to analyze cytokine levels of renal tubular epithelial cell lines (NRK-52E, HK2) and human renal glomerular endothelial cell line (hrGECs).UC-MSCs significantly ameliorated functional parameters, such as 24-h urinary protein, creatinine clearance rate, serum creatinine, urea nitrogen, and renal hypertrophy index. Pathological changes in the kidney were manifested by significant reductions in renal vacuole degeneration, inflammatory cell infiltration, and renal interstitial fibrosis after UC-MSC treatment. We observed that the number of UC-MSCs recruited to the injured kidneys was increased compared with the controls. UC-MSCs apparently reduced the levels of pro-inflammatory cytokines (IL-6, IL-1β, and TNF-α) and pro-fibrotic factor (TGF-β) in the kidney and blood of DN rats. In vitro experiments showed that UC-MSC conditioned medium and UC-MSC-derived exosomes decreased the production of these cytokines in high glucose-injured renal tubular epithelial cells, and renal glomerular endothelial cells. Moreover, UC-MSCs secreted large amounts of growth factors including epidermal growth factor, fibroblast growth factor, hepatocyte growth factor, and vascular endothelial growth factor.UC-MSCs can effectively improve the renal function, inhibit inflammation and fibrosis, and prevent its progression in a model of diabetes-induced chronic renal injury, indicating that UC-MSCs could be a promising treatment strategy for DN.
Endometrial reconstruction from stem cells
[J].Adult stem cells have been identified in the highly regenerative human endometrium on the basis of their functional attributes. They can reconstruct endometrial tissue in vivo suggesting their possible use in treating disorders associated with inadequate endometrium. The identification of specific markers for endometrial mesenchymal stem cells and candidate markers for epithelial progenitor cells enables the potential use of endometrial stem/progenitor cells in reconstructing endometrial tissue in Asherman syndrome and intrauterine adhesions.Copyright © 2012 American Society for Reproductive Medicine. Published by Elsevier Inc. All rights reserved.
Quantification of the CM-Dil-labeled human umbilical cord mesenchymal stem cells migrated to the dual injured uterus in SD rat
[J].
Umbilical cord-derived mesenchymal stem cells on scaffolds facilitate collagen degradation via upregulation of MMP-9 in rat uterine scars
[J].Background: Severe injuries of the uterus may trigger uterine scar formation, ultimately leading to infertility or obstetrical complications. To date, few methods have adequately solved the problem of collagen deposition in uterine scars. Umbilical cord-derived mesenchymal stem cells (UC-MSCs) have shown great promise in clinical applications. The objective of this study was to investigate the effect of a scaffold/UC-MSCs construct on collagen degradation and functional regeneration in rat uterine scars following full-thickness excision of uterine walls.Methods: In order to establish a rat model of uterine scars, the uterine wall of approximately 1.0 cm in length and 0.5 cm in width (one-third of the uterine circumference) was excised from each uterine horn. A total of 128 scarred uterine horns from 64 rats were randomly assigned to four groups, including a PBS group (n = 32 uterine horns), scaffold group (n = 32 uterine horns), UC-MSCs group (n = 32 uterine horns) and scaffold/UC-MSCs group (n = 32 uterine horns) to investigate the effect of different treatments on the structure and function of uterine scars. PBS, degradable collagen fibres, UC-MSCs or UC-MSCs mixed with gelatinous degradable collagen fibres were injected into four pre-marked points surrounding each uterine scar, respectively. At days 30 and 60 post-transplantation, a subset of rats (n = 8 uterine horns) from each group was euthanized and serial sections of uterine tissues containing the operative region were prepared. Haematoxylin-eosin staining, Masson's trichrome staining, and immunohistochemical staining for MMP-2, MMP-9, alpha-SMA and vWF were performed. Finally, another subset of rats (n = 16 uterine horns) from each group was mated with male rats at day 60 post-transplantation and euthanized 18 days after the presence of vaginal plugs to check numbers, sizes and weights of fetuses, as well as sites of implantation.Results: The scaffold/UC-MSCs group exhibited obvious collagen degradation compared with the other three groups. At day 60 post-transplantation, the number of MMP-9-positive cells in the scaffold/UC-MSCs group (25.96 +/- 3.63) was significantly higher than that in the PBS group (8.19 +/- 1.61, P < 0.01), the scaffold group (7.25 +/- 2.17, P < 0.01) and the UC-MSCs group (8.31 +/- 2.77, P < 0.01). The pregnancy rate in the scaffold/UC-MSCs group (10/16) was also significantly higher than that in the PBS group (2/16, P < 0.017), the scaffold group (1/16, P < 0.017) and the UC-MSCs group (3/16, P < 0.017).Conclusions: The scaffold/UC-MSCs system facilitated collagen degradation in uterine scars via upregulation of MMP-9, which was secreted by transplanted UC-MSCs, and promoted regeneration of the endometrium, myometrium and blood vessels in uterine scars. Furthermore, the scaffold/UC-MSCs-treated uterine scars showed nearly complete restoration of receptive fertility.
Allogeneic cell therapy using umbilical cord MSCs on collagen scaffolds for patients with recurrent uterine adhesion: a phaseⅠclinical trial
[J].
Human Umbilical Cord Mesenchymal Stem Cell-Derived Conditioned Medium Promotes Human Endometrial Cell Proliferation through Wnt/β-Catenin Signaling
[J].
Repair abilities of mouse autologous adipose-derived stem cells and ShakeGelTM3D complex local injection with intrauterine adhesion by BMP7-Smad5 signaling pathway activation
[J].
Emulating Human Tissues and Organs: A Bioprinting Perspective Toward Personalized Medicine
[J].
Bioprinting of a Blue Light-Cross-Linked Biodegradable Hydrogel Encapsulating Amniotic Mesenchymal Stem Cells for Intrauterine Adhesion Prevention
[J].Intrauterine adhesion (IUA) is a common and prevailing complication after uterine surgery, which can lead to clinical symptoms such as a low menstrual volume, amenorrhea, periodic lower abdominal pain, infertility, and so on. Placing a three-dimensional printing hydrogel between the injured site and the adjacent tissue is considered to be a physical barrier to prevent adhesion, which can isolate the damaged area during the healing process. In this work, a tissue hydrogel with various proportions of a methacrylated gelatin (GelMA) and methacrylated collagen (ColMA) composite hydrogel loaded with amniotic mesenchymal stem cells (AMSCs) was constructed by using three-dimensional biological printing technology. Compared with the single GelMA hydrogel, the composite antiadhesion hydrogel (GelMA/ColMA) showed an appropriate swelling ratio, enhanced mechanical properties, and impressive stability. Meanwhile, the microstructure of the GelMA/ColMA composite hydrogel showed a denser and interconnected microporous structure. In addition, the cytotoxicity study indicated that the GelMA/ColMA hydrogel has a cytocompatibility nature toward AMSCs. Finally, the fabrication of stem cell encapsulation hydrogels was studied, and the cells could be released continuously for more than 7 days with the normal cell function. The results of in vivo experiments indicated that the GelMA/ColMA/hAMSC (human amnion mesenchymal stem cell) hydrogel can prevent cavity adhesion in a rat IUA model. Therefore, bioprinting a biodegradable hydrogel cross-linked by blue light has satisfactory anticavity adhesion effects with excellent physical properties and biocompatibility, which could be used as a preventive barrier for intrauterine adhesion.© 2021 The Authors. Published by American Chemical Society.
Injectable 3D-Printed Porous Scaffolds for Adipose Stem Cell Delivery and Endometrial Regeneration
[J].
Adipose mesenchymal stem cell-derived exosomes promote cell proliferation, migration, and inhibit cell apoptosis via Wnt/β-catenin signaling in cutaneous wound healing
[J].Cutaneous wounds, a type of soft tissue injury, are difficult to heal in aging. Differentiation, migration, proliferation, and apoptosis of skin cells are identified as key factors during wound healing processes. Mesenchymal stem cells have been documented as possible candidates for wound healing treatment because their use could augment the regenerative capacity of many tissues. However, the effects of exosomes derived from adipose-derived stem cell (ADSC-exos) on cutaneous wound healing remain to be carefully elucidated. In this present study, HaCaT cells were exposed to hydrogen peroxide (H O ) for the establishment of the skin lesion model. Cell Counting Kit-8 assay, migration assay, and flow cytometry assay were conducted to detect the biological function of ADSC-exos in skin lesion model. Finally, the possible mechanism was further investigated using Western blot assay. The successful construction of the skin lesion model was confirmed by results of the enhanced cell apoptosis of HaCaT cells induced by H O, the increased Bax expression and decreased Bcl-2 expression. CD9 and CD63 expression evidenced the existence of ADSC-exos. The results of functional experiments demonstrated that ADSC-exos could prompt cell proliferation and migration of HaCaT cells, and repress cell apoptosis of HaCaT cells. In addition, the activation of Wnt/β-catenin signaling was confirmed by the enhanced expression of β-catenin at the protein level. Collectively, our findings suggest that ADSC-exos play a positive role in cutaneous wound healing possibly via Wnt/β-catenin signaling. Our study may provide new insights into the therapeutic target for cutaneous wound healing.© 2019 Wiley Periodicals, Inc.
Adipose-derived mesenchymal stem cell-derived exosomes alleviate overwhelming systemic inflammatory reaction and organ damage and improve outcome in rat sepsis syndrome
[J].
MicroRNA-29b Inhibits Endometrial Fibrosis by Regulating the Sp1-TGF-β1/Smad-CTGF Axis in a Rat Model
[J].Intrauterine adhesions (IUAs), which are characterized by endometrial fibrosis, increase the risk of secondary infertility and recurrent miscarriage. MicroRNA-29 (miR-29) is a potent inhibitor of TGF-β1/Smad signaling. In this study, we investigated the therapeutic potential of agomir-29b, an miR-29b mimic, in endometrial fibrosis induced by dual injury (uterine curettage and lipopolysaccharide treatment) in a rat model of IUA and explored the underlying mechanism. We found that injured rats developed endometrial fibrosis characterized by increased COL1A1 and α-smooth muscle actin expression and decreased E-cadherin expression, associated with a loss of miR-29b. Overexpression of miR-29b before injury prevented endometrial fibrosis including collagen accumulation and epithelial-mesenchymal transition. Delay of agomir-29b treatment until endometrial fibrosis was established on day 4 also halted the progression of disease. Further experiments indicated that miR-29b inhibited endometrial fibrosis via blockade of the Sp1-TGF-β1/Smad-CTGF pathway. In conclusion, agomir-29b may act as a novel and effective therapeutic agent against IUAs. © The Author(s) 2015.
Bone mesenchymal stem cells-derived miR-223-3p-containing exosomes ameliorate lipopolysaccharide-induced acute uterine injury via interacting with endothelial progenitor cells
[J].Bone mesenchymal stem cells (BMSCs) have been used for the treatment of acute uterine injury (AUI)-induced intrauterine adhesion (IUA) via interacting with the endothelial progenitor cells (EPCs), and BMSCs-derived exosomes (BMSCs-exo) may be the key regulators for this process. However, the underlying mechanisms have not been studied. Based on the existed literatures, lipopolysaccharide (LPS) was used to induce AUI in mice models and EPCs to mimic the realistic pathogenesis of IUA and. Our data suggested that LPS induced apoptotic and pyroptotic cell death in mice uterine horn tissues and EPCs, and the clinical data supported that increased levels of pro-inflammatory cytokines IL-18 and IL-1β were also observed in IUA patients' serum samples, and silencing of NLRP3 rescued cell viability in LPS-treated EPCs. Next, the LPS-treated EPCs were respectively co-cultured with BMSCs in the Transwell system and BMSCs-exo, and the results hinted that both BMSCs and BMSCs-exo reversed the promoting effects of LPS treatment-induced cell death in EPCs. Then, we screened out miR-223-3p, as the upstream regulator for NLRP3, was enriched in BMSCs-exo, and BMSCs-exo inactivated NLRP3-mediated cell pyroptosis in EPCs via delivering miR-223-3p. Interestingly, upregulation of miR-223-3p attenuated LPS-induced cell death in EPCs. Collectively, we concluded that BMSCs-exo upregulated miR-223-3p to degrade NLRP3 in EPCs, which further reversed the cytotoxic effects of LPS treatment on EPCs to ameliorate LPS-induced AUI.
Exosomes Derived from Adipose Mesenchymal Stem Cells Restore Functional Endometrium in a Rat Model of Intrauterine Adhesions
[J].Intrauterine adhesion (IUA) caused by endometrial injury is one of the important causes of infertility in women of reproductive age and requires advanced treatment strategies. Increasing evidence suggests that the therapeutic effects of mesenchymal stem cells (MSC) mainly depend on their capacity to secrete paracrine factors and are mediated by MSC-derived exosomes. This study aimed to identify exosomes derived from adipose-derived mesenchymal stem cells (ADSC-exo) and explore the therapeutic potential in IUA rat models. ADSC-exo exhibited classic cup-shaped morphology with a positive expression of Alix and CD63 and were mainly concentrated at 109.5 nm. In IUA model, treatment with ADSC-exo maintained normal uterine structure, promoted endometrial regeneration and collagen remodeling, and enhanced the expression of integrin-β3, LIF, and VEGF. An improved receptivity of the regenerated endometrium was confirmed. Our findings demonstrated that ADSC-exo promoted endometrial regeneration and fertility restoration. It suggested that topical administration of ADSC-exo in uterus could be a promising strategy for patients suffering severe intrauterine adhesions and infertility.
Human mesenchymal stem cell-derived extracellular vesicles/estrogen combined therapy safely ameliorates experimentally induced intrauterine adhesions in a female rat model
[J].Background: Mesenchymal stem cells (MSCs) have diverse functions in regulating injury and inflammation through the secretion of extracellular vesicles (EVs).Methods: In this study, we investigated the systemic administration of extracellular vesicles derived from human umbilical cord mesenchymal stem cells (UCMSCs-EVs) as a therapeutic agent for intrauterine adhesions (IUAs) caused by endometrial injury. Additionally, we investigated the therapeutic impact of both UCMSCs-EVs and estrogen either separately or in combination in a rat model. The inflammation, vascularization, proliferation, and extent of fibrosis were assessed by a histopathological and immunohistochemical assessment using transforming growth factor (TGF)-beta as a fibrotic marker and vascular endothelial growth factor (VEGF) as a vascular marker. Additionally, quantitative real-time polymerase chain reaction (qRT-PCR) was used to analyze the expression of tumor necrosis factor (TNF)-alpha, interleukin (IL)-1, IL-6 (inflammatory cytokines), CD140b (a marker of endometrial stem cells), and RUNX2 (an antifibrotic factor). Finally, Western blotting was used to evaluate collagen I and beta-actin expression.Results: The therapeutic groups treated with either UCMSCs-EVs alone or combined with estrogen exhibited a significant decrease in inflammation and fibrosis (TNF-alpha, TGF-beta, IL-1, IL-6, RUNX2, and collagen-I) as well as a significant decrease in vascularization (VEGF) compared with the untreated rats with IUAs. The most significant results were obtained in animals with IUAs that received a combined therapy of UCMSCs-EVs and estrogen.Conclusions: We conclude that the synergistic action of human UCMSCs-EVs combined with estrogen provides a highly effective alternative regenerative agent in IUA treatment.